|
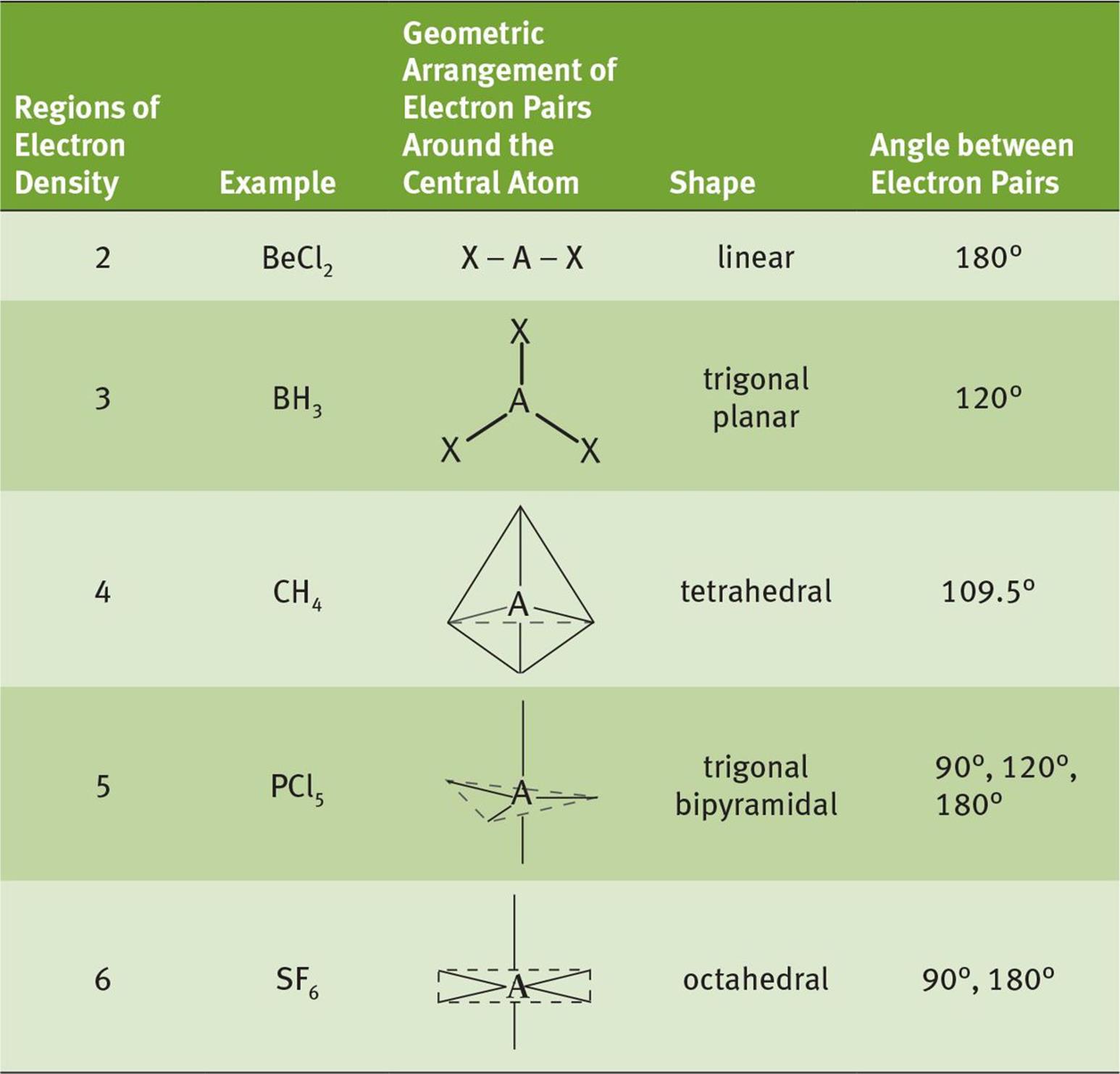
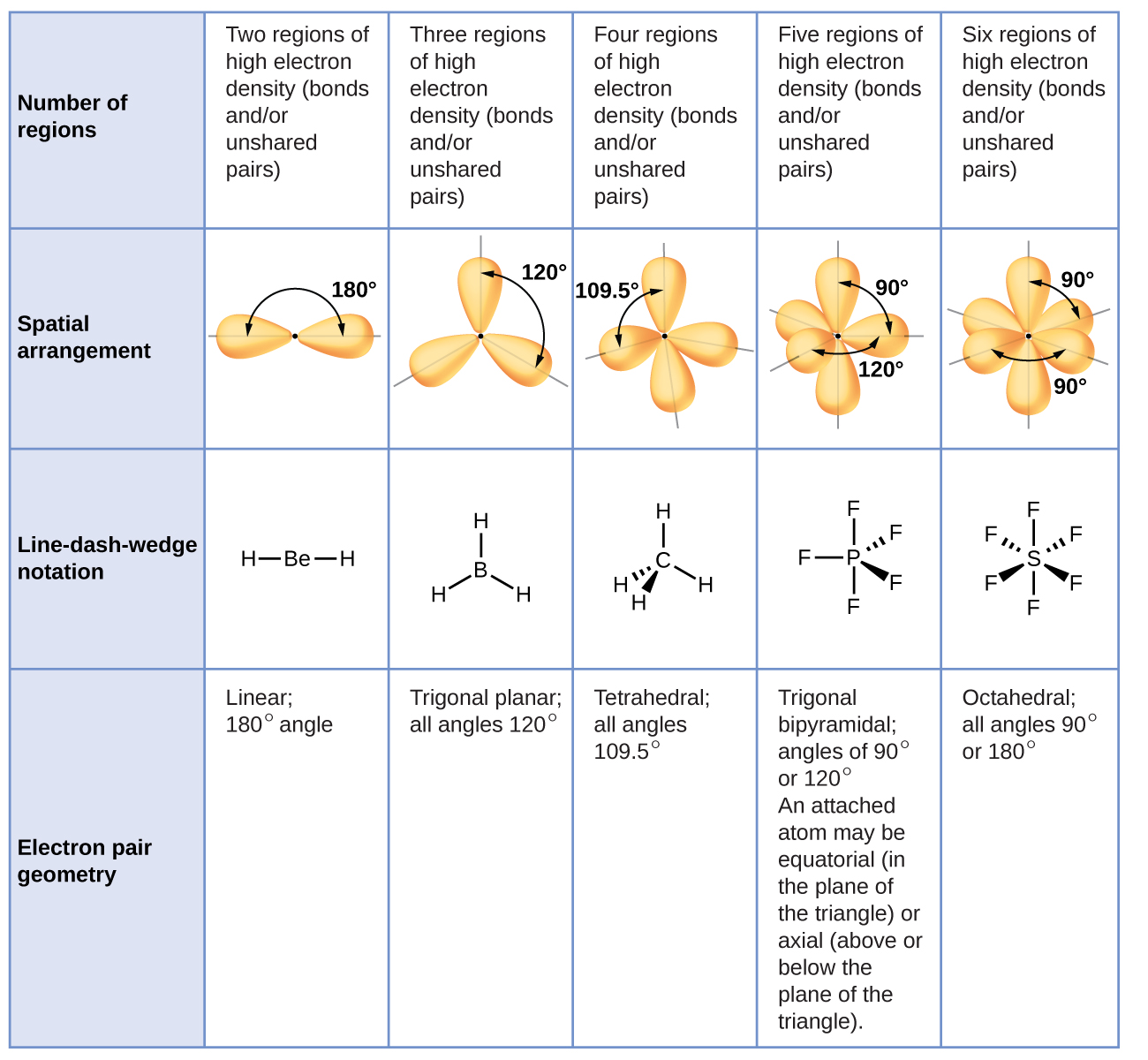
Check Your Learning Carbonate, CO 3 2, CO 3 2, is a common polyatomic ion found in various materials from eggshells to antacids. Note that the VSEPR geometry indicates the correct bond angles (120), unlike the Lewis structure shown above. For example, H 2 O has AX 2 E 2 formula so. The predicted ideal bond angle for AX 2 E 2 is <109.5º (104.5º). The electron geometry is tetrahedral and the shape is bent. For AX 2 E 2, the bond pair (X) is 2, and the lone pair (E) is also 2. It is a set of rules used in chemistry to predict the geometry of a molecule.\): Geometric structure of methyl isocyanate (CH 3–N=C=O), note there is no rotation around the double bonds only the single CN bond can rotate. The electron-pair geometry and molecular structure of BCl 3 are both trigonal planar. For instance, let’s say the VSEPR notation is AX 2 E 2. VSEPR theory stands for valence shell electron pair repulsion theory. Saturated Unsaturated and Supersaturated.We can draw the Lewis structure on a sheet of paper. Lets consider the Lewis structure for CCl 4. 
For bent molecular geometry when the electron-pair geometry is tetrahedral the bond angle is around 105 degrees. So the bongo will be the distance from here to here. The bond angle would be the distance from here to here. So if we take a look at this, we have here are central element and its connected to two surrounding elements. Reaction Quotient and Le Chatelier's Principle For trigonal pyramidal geometry the bond angle is slightly less than 109.5 degrees, around 107 degrees. a bond angle represents the angle between two bonds that begins from the same element within a molecule. 
The three fluorines contribute one electron. Chlorine is in group 7 and so has 7 outer electrons. The trigonal bipyramid therefore has two different bond angles - 120° and 90°.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
